|
As a result, a solvent such as ethanol or acetone, which is miscible with both silicon alkoxides and water, is added in order to get everything into the same phase so the necessary chemical reactions can occur. Because silicon alkoxides are usually non-polar liquids, however, they are not miscible with water. In this technique, a silicon alkoxide (usually either tetramethoxysilane or tetraethoxysilane) serves as the source for the silica, water acts as a reactant to help join the alkoxide molecules together, and a catalyst (such as ammonium hydroxide or ammonium fluoride) helps the underlying chemical reactions go fast enough to be practically useful. The most common technique used for producing silica gels today involves the reaction of a silicon alkoxide with water in a solvent such as ethanol or acetone, usually in the presence of basic, acidic, and/or fluoride-containing catalyst. 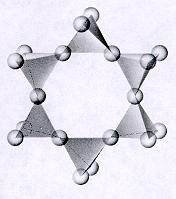
The Most Common Technique: Silicon Alkoxide Gelation See How Aerogel is Made for more information. Silica gels are produced through the sol-gel process, in which nanoparticles suspended in a liquid solution (i.e., a sol) are invoked to interconnect and form a continuous, porous, nanostructured network of particles across the volume of the liquid medium (i.e., a gel). This is typically done by supercritically drying the gel, but can also be done a number of other ways (see How Aerogel is Made). Silica aerogel is made by extracting the liquid from the framework of the silica gel in a way that preserves at least 50% (but typically 90-99+%) of the gel framework’s original volume. Silica gels are composed of two components-a solid, nanoporous silica-based framework which gives the gel its rigidity and solid form, and a liquid which permeates the pores of the framework. Silica gels used for preparing silica aerogels, on the other hand, are wet gels close to gelatin dessert in consistency (but a bit crumblier). It is important to note that the “silica gel” which comes in little packets marked “SILICA GEL DO NOT EAT” (usually found in consumer electronics packaging) is actually pellets of silica xerogel-a dense, dry, solid form of silica used to absorb moisture. A silica gel serves as the precursor in this case and can be prepared through a number of chemical processes. Like most other aerogels, silica aerogel starts its life out as a gel (see How Aerogel is Made). Here’s a dumb pneumonic to help keep it straight: when you hear silicA think glAss, when you hear silicOn think cOmputers, and when you hear siliconE think rubbEr. This said, if someone says “silicone aerogel” they probably just mean “silica aerogel”. Silicone aerogels can be and have been prepared and possess many properties characteristic of silicone rubbers. Think implants and temperature-resistant rubber. They are rubbery solids or liquids at room temperature and are very different from both silica and silicon. 
Silicones, on yet another hand, are polymers composed of silicon and oxygen, usually containing carbon and hydrogen as well. There are no reports of silicon aerogels yet, but they would certainly be interesting materials! They are very different! Unfortunately, you will see “silicon aerogel” mentioned in the media from time to time (even on one of NASA’s websites!), however this is very incorrect, and should instead read “silica aerogel” (even ’s own co-founder Will Walker confuses the terms from time to time, but we forgive him). Silicon is a semiconductor metalloid used in microchips, whereas silica is an insulating glassy material. Silica refers to the oxide of silicon, which has the empirical formula SiO 2. If the net weight corresponds to an amount greater than the PEL value after considering the associated SAE, the sample may be analyzed for the applicable component and appropriate results will be reported.įOR TOTAL DUST SAMPLES, DO NOT USE A CYCLONE!!Īll sampling instructions above are recommended guidelines for OSHA Compliance Safety and Health Officers (CSHOs), please see the corresponding OSHA method reference for complete details.Silica Aerogel on a Hand A Note About Silica vs. If the net weight of the sample yields a concentration below the PEL after considering the associated SAE, the SLTC will perform no further work on the sample and the sample air concentration will be reported as the calculated gravimetric air concentration. Tared low-ash 37 mm PVC filter, 5 microns S122 (total) request using analyte code 9135
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
